Human Type I IFN Neutralizing Antibody Mixture
Catalog Number: 39000
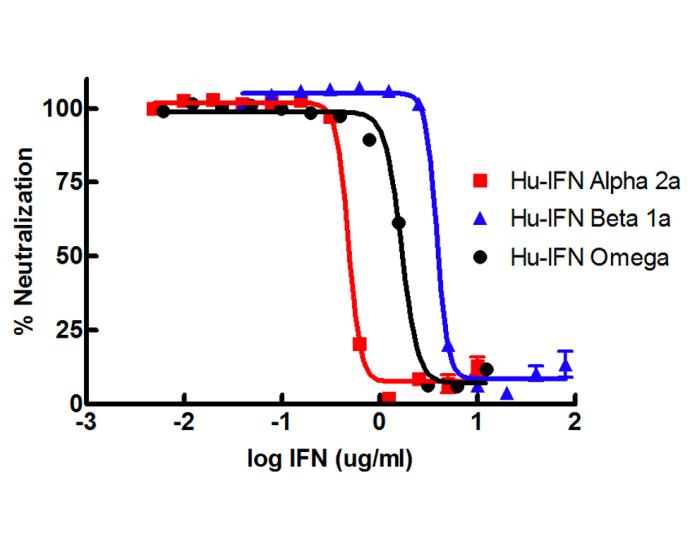
Highly recommended unconjugated mixture of MAbs and PAbs directed against human Type I IFN receptor and several Type I IFNs. Effectively neutralizes biological activity of human IFN-Alpha, IFN-Beta, IFN-Omega, IFN-Kappa, and IFN-Epsilon.
$700.00
Product Info
Blocks biological activity of human Type I interferons (alpha, beta, omega, kappa, epsilon)
Specificity
No cross-reactivity observed with the following:
Tested Application
Improves neutralization of multiple Type I IFNs often found in complex samples by employing a broadly neutralizing custom antibody cocktail.
Specifications
| Formulation | Supplied frozen in phosphate-buffered saline (PBS) |
|---|---|
| Purification | A combination of Protein G affinity chromatography and other conventional chromatographical methods. Endotoxin level < 1 EU/μg |
| Bioactivity | Encephalomyocarditis Virus (EMCV) was used to challenge A549 cells which were incubated with a constant concentration of the antibody mixture and the titrated interferon. |
| Storage | For retention of full activity store at -70°C or below and avoid repeated freeze/thaw cycles |
| Synonyms | Type I interferon, Type I IFN, Neutralizing, Neutralization, Type I Interferon Receptor, Antibody Cocktail |
Citations
7 Citations
- Lim, H. A. et al., (2023), "Innate antiviral responses against Shaan virus infection in HEK293, A549 and MARC-145 cells and limited role of viperin against Shaan virus replication", Heliyon, 9:e22597, DOI: 10.1016/j.heliyon.2023.e22597 (link)
- Tabtieng, T. et al., (2022), "Caspase-Mediated Regulation and Cellular Heterogeneity of the cGAS/STING Pathway in Kaposi's Sarcoma-Associated Herpesvirus Infection", mBio, e:0244622, PMID: 36255240, DOI: 10.1128/mbio.02446-22 (link)
- Tsuji, S. et al., (2022), "SARS-CoV-2 infection triggers paracrine senescence and leads to a sustained senescence-associated inflammatory response", Nature Aging, DOI: 10.1038/s43587-022-00170-7, (link)
- Essaidi-Laziosi, M., et al., (2021), "Sequential infections with rhinovirus and influenza modulate the replicative capacity of SARS-CoV-2 in the upper respiratory tract", Emerging Microbes & Infections, DOI: 10.1080/22221751.2021.2021806 (link)
- Lundberg, Rickard, et al. (2019). Zika Virus Non-Structural Protein NS5 Inhibits the RIG-I Pathway and Interferon Lambda 1 Promoter Activation by Targeting IKK Epsilon. Viruses, 20 pgs. PMID: 31690057. (link)
- Szabo, Peter, et al. (2019). Single-cell transcriptomics of human T cells reveals tissue and activation signatures in health and disease. Nature Communications, 16 pgs. PMID: 31624246. (link)
- Kuse, Nozomi, et al. (2019). Priming of HIV-1-specific CD8+ T cells with strong functional properties from naïve T cells. EBioMedicine, 11 pgs. PMID: 30956171. (link)