Abstract:
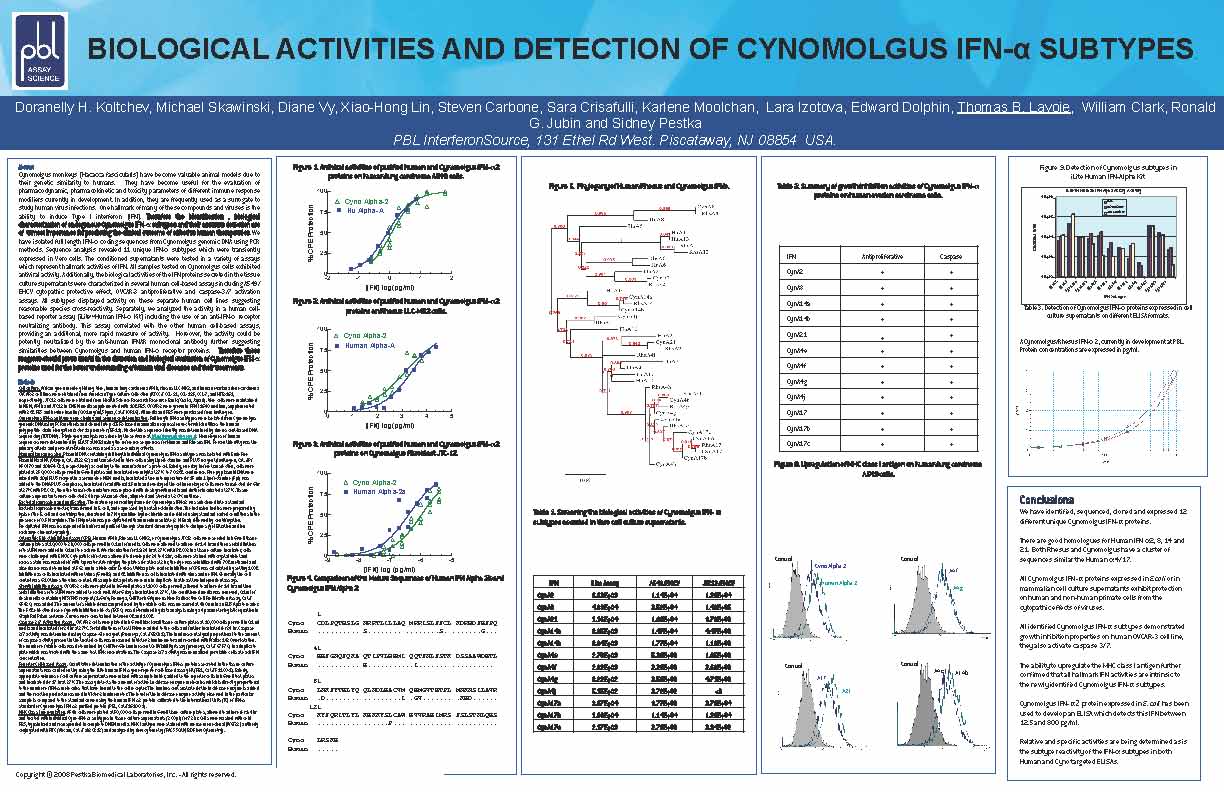
Cynomolgus monkeys (Macacca fascicularis) have become valuable animal models due to their genetic similarity to humans. They have become useful for the evaluation of pharmacodynamic, pharmacokinetic and toxicity parameters of different immune response modifiers currently in development. In addition, they are frequently used as a surrogate to study human virus infections. One hallmark of many of these compounds and viruses is the ability to induce Type I Interferon (IFN). Therefore the identification, biological characterization of endogenous Cynomolgus IFN-α subtypes and their accurate detection are of utmost importance for predicting the clinical outcome of effective human therapeutics. We have isolated full length IFN-α coding sequences from Cynomolgus genomic DNA using PCR methods. Sequence analysis revealed 11 unique IFN-α subtypes which were transiently expressed in Vero cells. The conditioned supernatants were tested in a variety of assays which represent hallmark activities of IFN. All samples tested on Cynomolgus cells exhibited antiviral activity. Additionally, the biological activities of the IFN proteins secreted in the tissue culture supernatants were characterized in several human cell-based assays including A549/EMCV cytopathic protective effect, OVCAR-3 antiproliferative and caspase-3/7 activation assays. All subtypes displayed activity on these separate human cell lines suggesting reasonable species cross-reactivity. Separately, we analyzed the activity in a human cell-based receptor assay (iLite Human IFN-α kit) including the use of an anti-IFN-α receptor neutralizing antibody. This assay correlated with the other human cell-line assays, providing an additional, more rapid measure of activity. Moreover, the activity could be potently neutralized by the anti-human IFNAR monoclonal antibody further suggesting similarities between Cynomolgus and human IFN-α receptor proteins. Therefore these reagents should prove useful in the detection and biological evaluation of Cynomolgus IFN-α proteins for the better understanding of human viral disease and their treatments.