Types I, II, and III of Interferon
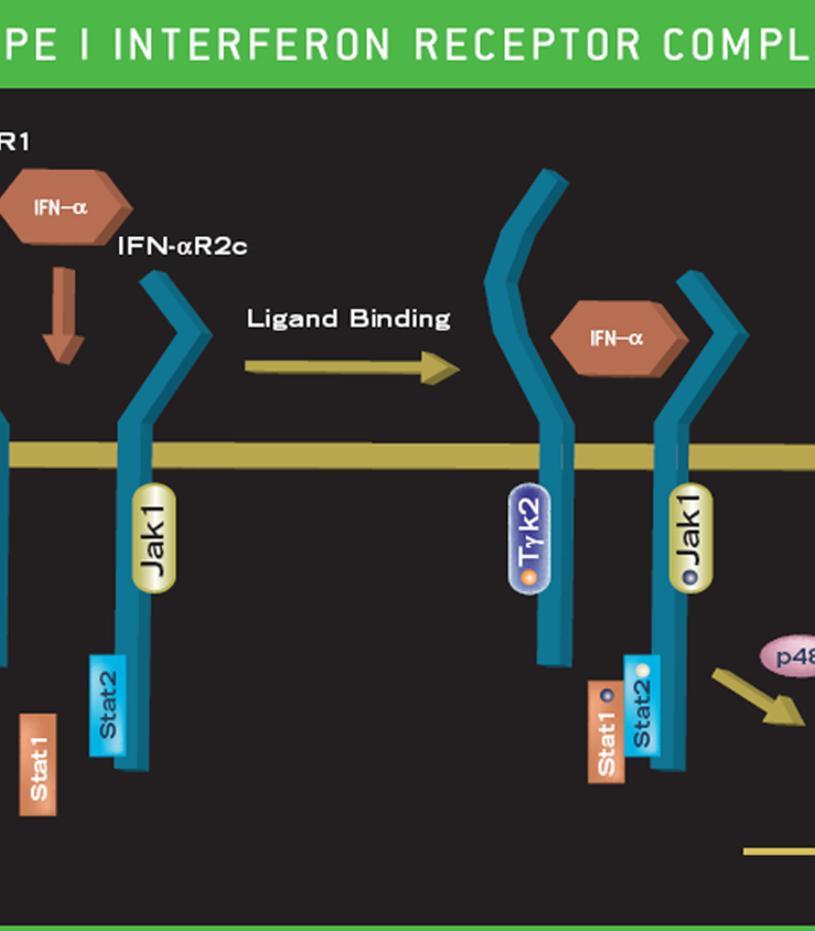
Mammalian type I IFNs consist of over nine distinct classes that include IFN-α, IFN-β, IFN-δ, IFN-ε, IFN-κ, IFN-ω, IFN-υ, IFN-τ and IFN-ζ. While IFN-α, IFN-β, IFN-ε, IFN-κ, IFN-ω, and IFN-υ are found in humans, IFN-δ, IFN-τ and IFN-ζ are not. These IFNs bind to the type I receptor which is composed of two chains, commonly designated IFNAR1 and IFNAR2. Type I IFNs are typically produced by macrophages, neutrophils, dendritic cells and other somatic cells in response to many viruses and some pathogens.
In humans, IFN-α consists of a group of proteins that are greater than 85% homologous by amino acid sequence. Only one human IFN-α is N-glycosylated and a few IFN-α species have been shown to be O-glycosylated. In the mouse, nearly all of the IFN-α species are N-glycosylated. IFN-β, is produced by a variety of cells in response to viral challenge, and the native human IFN-β bears a single N-glycosylation site. The other type I IFNs have not been studied extensively as IFN-α and IFN-β. Type II IFN in humans is limited to a single IFN-γ gene. This IFN binds to the Type II receptor comprised of IFNGR1 (IFN-γR1) and IFNGR2 (IFN-γR2) chains. IFN-γ is produced by cells of the immune system such as T-cells and NK cells. IFN-γ is glycosylated in mammalian cells, and functions as a homodimer. On a mass basis, IFN-α and IFN-β exhibit more potent antiviral activity than IFN-γ.
Often, the expression of the type I IFNs is induced by engagement of Toll-Like Receptors (TLR). The innate immune system has evolved the ability to recognize non-self motifs through the TLR receptors, e.g., double stranded RNAs through TLR3, lipopolysaccharides through TLR4, and methylated CpG DNA motifs through TLR9. Interferon produced by TLR-activated cells can function in an autocrine or paracrine manner limiting pathogen infection. When IFN interacts with its cognate receptor, a signal is rapidly transmitted within the cell, often producing an antiviral state. The primary signal transduction cascade promoted by type I IFNs is the JAK1-STAT pathway (see above).
Activation of this signal transduction pathway leads to increased gene expression including (2’-5’) oligoadenylate synthetases, Mx proteins, and protein kinase R (PKR) that protect the cell from viral infection. In fact, a host of genes are expressed in response to interferons many of which have roles yet to be determined.
It remains unclear why there are so many different Type I IFNs including multiple IFN-α subtypes. A variety of studies suggested they possess overlapping but also unique sets of biological activities. Additional studies are revealing that type I IFNs may also play immunoregulatory roles. In contrast, the primary role of IFN-γ is the activation and development of adaptive immune functions with a lesser role in innate immune responses.
Type III interferons [IFN-λ1 (IL-29), IFN-λ2 (IL-28A), and IFN-λ3 (IL-28B)] are newly identified class II cytokine receptor ligands that are distantly related to members of the IL-10 family (11-13% amino acid sequence identity) and type I IFN family (15-19% amino acid sequence identity). All three cytokines exert bioactivities that overlap those of type I IFNs, including antiviral activity and up-regulation of MHC class I antigen expression. The three proteins signal through the same heterodimeric receptor complex that is composed of the IL-10 receptor β (IL-10Rβ) and a novel IL-28 receptor α (IL-28Rα, also known as IFN-λ R1). Ligand binding to the receptor complex induces JAK activation and STAT1/STAT2 tyrosine phosphorylation. The phosphorylated STAT1 and STAT2 combine with IFN-regulatory factor 9 (IRF-9) to form IFN-stimulated regulatory factor 3 (ISGF-3) transcription factor complex that translocates to the nucleus. Recent gene knockout studies (Ank et al.2006. J. Interferon Cytokine Res. 26:373; Ank et al. 2008. J. Immunol. 180:2474) suggest that type III IFN signaling pathway may have been conserved to combat pathogens that target type I IFN signaling pathways.
Types I, II, and III of Interferon
Mammalian type I IFNs consist of over nine distinct classes that include IFN-α, IFN-β, IFN-δ, IFN-ε, IFN-κ, IFN-ω, IFN-υ, IFN-τ and IFN-ζ. While IFN-α, IFN-β, IFN-ε, IFN-κ, IFN-ω, and IFN-υ are found in humans, IFN-δ, IFN-τ and IFN-ζ are not. These IFNs bind to the type I receptor which is composed of two chains, commonly designated IFNAR1 and IFNAR2. Type I IFNs are typically produced by macrophages, neutrophils, dendritic cells and other somatic cells in response to many viruses and some pathogens.
In humans, IFN-α consists of a group of proteins that are greater than 85% homologous by amino acid sequence. Only one human IFN-α is N-glycosylated and a few IFN-α species have been shown to be O-glycosylated. In the mouse, nearly all of the IFN-α species are N-glycosylated. IFN-β, is produced by a variety of cells in response to viral challenge, and the native human IFN-β bears a single N-glycosylation site. The other type I IFNs have not been studied extensively as IFN-α and IFN-β. Type II IFN in humans is limited to a single IFN-γ gene. This IFN binds to the Type II receptor comprised of IFNGR1 (IFN-γR1) and IFNGR2 (IFN-γR2) chains. IFN-γ is produced by cells of the immune system such as T-cells and NK cells. IFN-γ is glycosylated in mammalian cells, and functions as a homodimer. On a mass basis, IFN-α and IFN-β exhibit more potent antiviral activity than IFN-γ.
Often, the expression of the type I IFNs is induced by engagement of Toll-Like Receptors (TLR). The innate immune system has evolved the ability to recognize non-self motifs through the TLR receptors, e.g., double stranded RNAs through TLR3, lipopolysaccharides through TLR4, and methylated CpG DNA motifs through TLR9. Interferon produced by TLR-activated cells can function in an autocrine or paracrine manner limiting pathogen infection. When IFN interacts with its cognate receptor, a signal is rapidly transmitted within the cell, often producing an antiviral state. The primary signal transduction cascade promoted by type I IFNs is the JAK1-STAT pathway (see above).
Activation of this signal transduction pathway leads to increased gene expression including (2’-5’) oligoadenylate synthetases, Mx proteins, and protein kinase R (PKR) that protect the cell from viral infection. In fact, a host of genes are expressed in response to interferons many of which have roles yet to be determined.
It remains unclear why there are so many different Type I IFNs including multiple IFN-α subtypes. A variety of studies suggested they possess overlapping but also unique sets of biological activities. Additional studies are revealing that type I IFNs may also play immunoregulatory roles. In contrast, the primary role of IFN-γ is the activation and development of adaptive immune functions with a lesser role in innate immune responses.
Type III interferons [IFN-λ1 (IL-29), IFN-λ2 (IL-28A), and IFN-λ3 (IL-28B)] are newly identified class II cytokine receptor ligands that are distantly related to members of the IL-10 family (11-13% amino acid sequence identity) and type I IFN family (15-19% amino acid sequence identity). All three cytokines exert bioactivities that overlap those of type I IFNs, including antiviral activity and up-regulation of MHC class I antigen expression. The three proteins signal through the same heterodimeric receptor complex that is composed of the IL-10 receptor β (IL-10Rβ) and a novel IL-28 receptor α (IL-28Rα, also known as IFN-λ R1). Ligand binding to the receptor complex induces JAK activation and STAT1/STAT2 tyrosine phosphorylation. The phosphorylated STAT1 and STAT2 combine with IFN-regulatory factor 9 (IRF-9) to form IFN-stimulated regulatory factor 3 (ISGF-3) transcription factor complex that translocates to the nucleus. Recent gene knockout studies (Ank et al.2006. J. Interferon Cytokine Res. 26:373; Ank et al. 2008. J. Immunol. 180:2474) suggest that type III IFN signaling pathway may have been conserved to combat pathogens that target type I IFN signaling pathways.