PBL can expedite your R&D work as our technologies and services can be tailored to meet your specific requirements under fit-for-purpose guidelines. For many of your sample testing and screening needs, PBL can help you measure basal and elevated levels of cytokine or biomarker in samples from healthy and disease states. With state-of-the-art technologies quantifying into femtogram/milliliter level of a biomarker to cost-effective multiplex profiling of inflammatory cytokines, PBL's immunoassay testing service offers a multitude of options to fit your project needs.
We understand the value of your experimental samples and execute assays as you would-consuming a minimum of sample while generating robust results. We can take on work you don't have the capacity to handle. Let us help you develop, customize, or just execute assays on your sample in a timely, professional manner.
MSD-ECL Assays
Meso Scale Discovery® Electrochemiluminescence (MESO QuickPlex SQ 120MM)
-
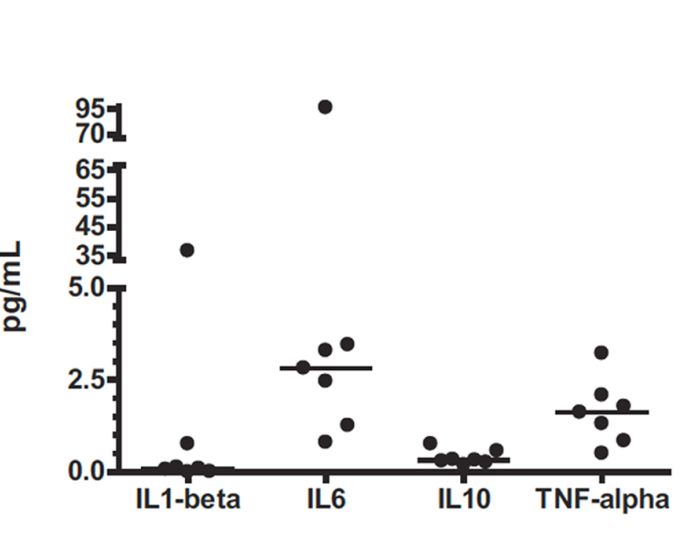
Measure high and low abundance analytes (normal and elevated levels) within the same sample with 5-Log+ dynamic range
-
180+ validated fit-for-purpose singleplex and preconfigured multiplex panel ELISAs (V-PLEX) available with excellent performance specifications and lot-to-lot consistency
Offering high-precision protein quantification services for a broad range of sample matrices for Human, Non-Human Primate (NHP), Mouse, Rat, and Canine samples. MSD's validated multiplex panel assay kits (V-PLEX) enable simultaneous comparison of the expression of pertinent cytokines, chemokines, and other biomarkers in samples from diseased and normal patients at different intervals.
Multiplex immunoassays on the MSD platform maintain the sensitivity and performance offered by singleplex ELISAs while providing additional benefits such as cost-savings and targeting of several related analytes in a single sample. The ability to quantify compatible analytes while requiring no more than 25 μl of neat sample ensures efficient use of precious sample.
Broad Range of Applications
The unique detection technology provided by the MSD platform utilizes SULFO-TAG™ labels which emit light upon electrochemical stimulation. This system provides the basis for achieving measurement of a several log range of protein expression levels.
-
Cytokines & Chemokines, Cardiac Biomarkers, Immunogenicity, Immunology, Inflammation, Intracellular signaling, Metabolic, Neurodegeneration, Oncology and Cancer, Toxicity, ...and more
Ultra-Sensitive Cytokine Testing
MilliporeSigma Single Molecule Counting (SMCxPRO®, Erenna®)
-
Sub-pg/ml sensitivity of low abundance biomarkers in normal or disease sera/plasma
-
Minimal sample dilution with 4-Log+ dynamic range
-
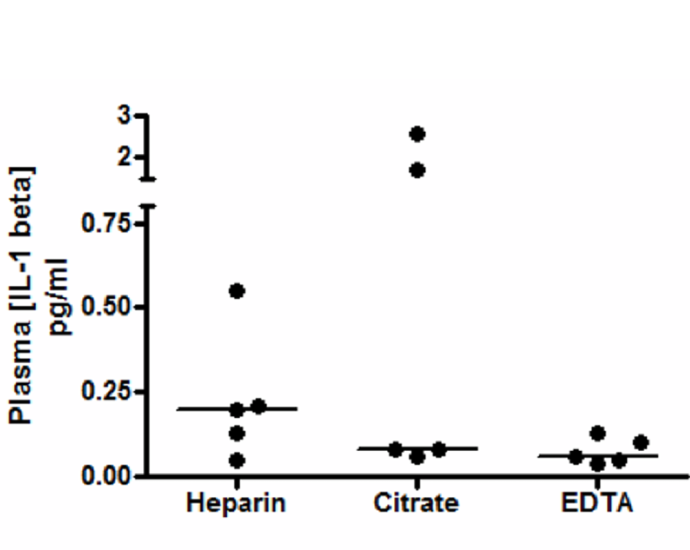
40+ cytokine assays covering several disease areas including IFN-γ, IL-1a, IL-1b, IL-4, IL-6, IL-8, IL-10, IL-13, IL-15, IL-17A, IL-17F, IL-17 heterodimer, IL-23, GLP-1, and TNF-α
MilliporeSigma's SMC (Single Molecule Counting) technology in conjunction with robust microparticle (MP) based sandwich immunoassays enables accurate, high-precision baseline quantification of low-abundance analytes at or below sub-picogram concentrations. Accurately measuring low-abundance analytes present in complex matrices is essential for the profiling and characterization of cytokines and other biomarker analytes.
An increased understanding of the role and regulation of cytokines in disease states results from greater profiling and characterization of their activities in biological responses. This technology can be instrumental in furthering our collective understanding of low-abundance cytokines in complex matrices.
Powered by SMCxPRO® & Erenna® Immunoassay Systems, MilliporeSigma Inc., Burlington, MA, USA.
Ultra-Sensitive Biomarker Testing
Quanterix Simoa (Quanterix HD-X, Quanterix SP-X)
-
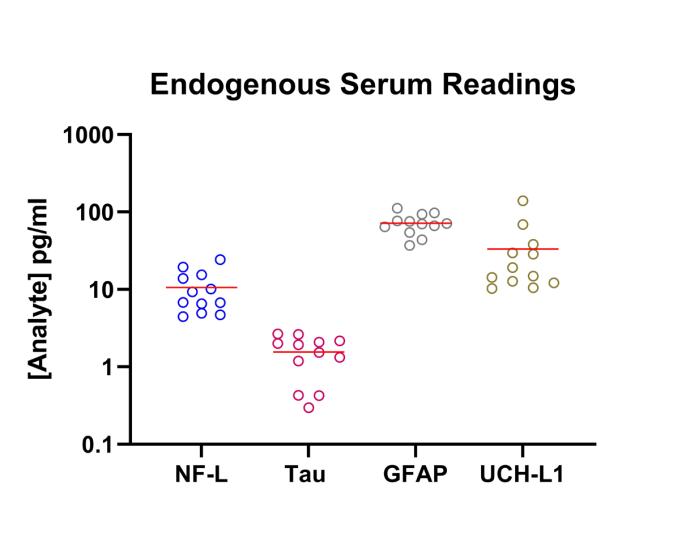
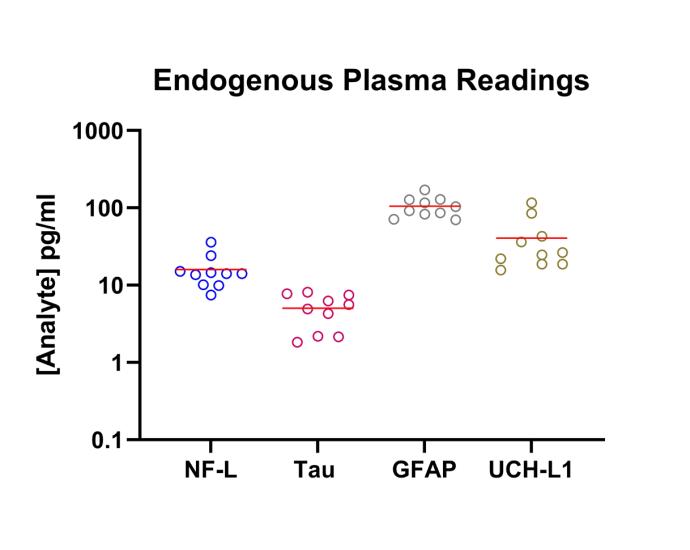
Femtogram per milliliter (fg/ml) level sensitivity for measuring low-abundance biomarkers in serum, plasma, and other complex matrices (e.g. bronchial lavage, urine, sputum)
-
50+ analyte panels available including IL-17A, TNF-alpha, IL-23, IP-10, TRAIL, IL-6, IL-10, and IL-12p70
Taking advantage of the benefits of Simoa (Single Molecule Array) technology, services run on this platform yield robust and reproducible data thus enabling a better understanding of the contributions of difficult-to-measure analytes. This technology allows for “digital” measurement of each captured bead for the presence of the targeted analyte in a sandwich immunoassay format. The enzymatic amplification of analyte binding vents on single beads in single wells yields low background signals and can increase assay sensitivities up to several orders of magnitude over conventional assays. Assays run on this platform frequently exhibit sensitivities in the low to mid fg/ml range.
The instrument’s capacity for digital measurement at low concentrations of target analyte and analog measurement at higher analyte concentrations ensures accuracy and precision across a wide dynamic range (>4 logs) minimizing sample volume requirements and the need for multiple dilutions. Simoa facilitates quantification of biomarkers at unprecedented levels.
Powered by SimoaTM (Single Molecule Array) Technology, Quanterix, Lexington, MA, USA.
NF-light® is a registered trademark of Uman Diagnostics, Umeå Sweden.
Single Analyte & Multiplex Assay Services
Single Analyte ELISA Service
Sensitive detection and robust results. Dedicated, flexible workforce. Reliable, high-quality outcomes.
Take advantage of PBL ELISAs or other commercial ELISAs for a wide range of additional analytes using PBL's services.
PBL ELISAs:
-
High sensitivity ELISAs available for a comprehensive range of species and analytes
-
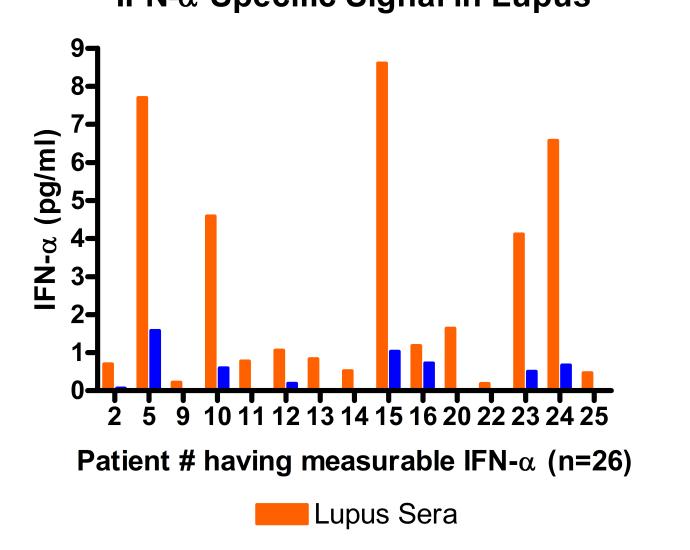
Matrix tolerance for autoimmune disease sera*
PBL routinely runs ELISA studies for our clients for the detection and differentiation of interferons and cytokines in various sample matrices. We offer a range of heterogeneous immunoassay services for:
- Human IFN-Alpha, Beta, Gamma, Lambda 1/2/3, Omega, Gamma Receptor 1
- Mouse IFN-Alpha, Beta, Lambda 2/3
- Cynomolgus/Rhesus IFN-Alpha, Cynomolgus IFN-Beta
- Porcine IFN-Alpha
Multiplex Screening Service
High sensitivity & specificity using low sample volumes. Cost-effective quantitation of multiple analytes.
-
Variety of chemokine and cytokine panels already available for human and mouse targets
-
Cost-effective multiplex protein biomarker screening using chemiluminescent detection
These ELISAs provide chemiluminescent results on each analyte and allow for a global understanding of ongoing immune responses in diseases ranging from cancer to autoimmunity. Evaluation of one given inflammatory molecule in the context of several others, repeated measurements of the same cytokine panels in the same subjects under different treatment assay conditions, and reliable detection of different proteins across a broad range of concentrations are just some of the benefits provided by this multiplex service.
-
Ability to run immunoassays produced by other suppliers, provided we are confident in the data they generate (contact PBL for your specific needs)
Talk to a Scientist